INTRODUCTION
Historical Context and the Birth of Modern Plasticization
Plasticizers in Personal Care Products: From 19th-Century Billiard Balls to Enhanced Tactile Performance and Skin Penetration
The concept of plasticization traces its origins to the 1860s, during a period of acute global ivory shortage caused by the booming popularity of billiards in Europe and North America. Traditional billiard balls were carved from elephant ivory, and besides its obvious and unnecessary brutality, supply could no longer meet demand. In 1863, the American inventor John Wesley Hyatt began experimenting with nitrocellulose, a rigid and brittle polymer. Pure nitrocellulose, however, was far too stiff and explosive for practical molding.
Hyatt’s breakthrough, patented in 1869–1870, was the addition of camphor as a plasticizer.[1] The resulting material, trademarked Celluloid, could be softened with heat and solvent, molded under pressure into perfect spheres, and cooled, and hardened into a tough, glossy product. Celluloid became the first commercially successful thermoplastic and ushered in the plastics/plasticizer era.
The principle discovered by Hyatt, that certain additives can dramatically enhance polymer chain mobility without chemically altering the base polymer, rapidly found applications far beyond billiards. By the early 20th century, a number of chemistries emerged for different matrices and applications, the most demonstrative among them being PVC where a rigid construction material (e.g. window frames) can be made into a soft and stretchy film (e.g. pool liners). Eventually, these plasticizers made their way into personal care products.
Today: Basic Necessities in Skincare
Modern personal care products are expected to deliver exceptionally high sensory and performance standards: uniform texture, lasting physical stability, a non-tacky finish, and a luxurious, smooth, creamy skin feel that consumers immediately associate with premium quality. Achieving these characteristics is far from trivial when one considers the complex, multicomponent nature of typical formulations. These systems routinely combine high-molecular-weight waxes (film-formers, thickeners), inorganic pigments and fillers, volatile and non-volatile oils, surfactants, fragrances, preservatives, active ingredients, and water, each contributing with different and sometimes conflicting physical properties.
Without careful rheological and interfacial control, the final product can become (excessively) brittle, prone to phase separation, or exhibit cracking upon drying. Films may lack flexibility, resulting in flaking, or become uneven, emulsions may feel greasy or sticky. All or any of these defects can directly translate into reduced consumer acceptance.
In the specific realm of skincare, particularly moisturizing products, efficacy historically revolves around three well-established categories:
- Emollients: lipophilic substances that “repair” the skin surface by filling intercellular spaces and micro fissures in the stratum corneum, restoring smoothness and flexibility.[2]
- Occlusives: materials that form a continuous hydrophobic film on the skin surface, dramatically reducing trans epidermal water loss (TEWL), basically a physical barrier.[3]
- Humectants: molecules with some hygroscopicity, capable of binding water from the environment (or deeper skin layers, i.e. Epidermis/Dermis) and delivering it to the Stratum Corneum.[4]
For true emollient performance, the material must exhibit sufficiently low viscosity and favorable wetting properties to flow rapidly into microscopic skin crevices via capillary action. This process is governed by Young–Laplace capillary pressure, surface tension (interfacial) tension, contact angle, and bulk viscosity, parameters that are strongly temperature-dependent. Many oils, waxes, or high molecular weight (wax) esters are either too viscous at room temperature or crystallize upon cooling, severely limiting their ability to penetrate and “repair” the skin surface effectively.
The classical solution to this dilemma has long been the careful selection and incorporation of a plasticizer. By selecting an appropriate plasticizer system, the scientist can also achieve additional functional benefits, which will be discussed later in this paper.
Defining Plasticizers in a Cosmetic Context
Etymologically, the term “plasticizer” derives from the Greek πλαστικός (plastikos), meaning “capable of being molded.” In polymer science, a plasticizer is formally defined as “a substance incorporated into a polymer system to increase its workability, flexibility, or distensibility by lowering the glasstransition temperature and elastic modulus of the material.”[5]
In personal care formulations, plasticizers perform multiple critical functions. They suppress crystallization and reduce melt and solution viscosity, which facilitates mixing, pumping, and filling operations during manufacturing. They also lower the glass transition temperature of filmforming materials, preventing brittle or cracking films after solvent evaporation when applicable. In addition, plasticizers enhance low-temperature flexibility of occlusive barriers, helping maintain a soft, non-waxy sensory profile even in cold environments. By reducing the yield stress of emulsions and anhydrous balms, they contribute to greater sensory elegance, including improved cushion, extended playtime, and reduced drag. Plasticizers further promote deeper and more uniform penetration of emollients into the stratum corneum by decreasing the viscosity, intermolecular cohesion, and surface tension of the oil phase.
| Plasticizer Function | Mechanistic Effect | Outcome / Benefit |
| Suppress crystallization & reduce viscosity | Decreases solid formation and eases flow | Facilitates mixing, pumping, and filling |
| Lower glass transition temperature | Increases flexibility of polymer/ film | Prevents brittle or cracking films after solvent evaporation |
| Enhance low-temperature flexibility | Maintains material softness | Keeps occlusive barriers soft, non-waxy in cold climates |
| Reduce yield stress | Lowers resistance in emulsions/balms | Improves sensory elegance, cushion, playtime, non-drag |
| Promote deeper emollient penetration | Decreases viscosity, cohesion, and surface tension | Ensures uniform penetration into stratum corneum |
The two main categories of plasticizers are primary and secondary. Primary plasticizers interact directly with wax molecules, whereas secondary plasticizers enhance the effectiveness of a primary plasticizer. Primary plasticizers can act via two mechanisms: internal or external plasticization. Internal plasticization involves chemical modification of the wax molecule or its building blocks before the wax is synthesized. External plasticization, which is the focus of this publication, occurs when plasticizers are added during formulation at elevated temperatures. Esters are among the most commonly used external plasticizers due to their favorable physical interactions with the high molecular weight molecules that typically constitute waxes. These interactions allow the wax and plasticizer to form a homogeneous physical unit, preventing phase separation. It is critical that the intermolecular interactions between wax molecules, typically long-chain hydrocarbons or esters, and plasticizer molecules are carefully balanced within an intermediate range. Interactions that are too strong or too weak can compromise the desired macroscopic properties of the final material. This specific interaction between the wax and plasticizer molecules causes these materials to form a homogenous physical unit, i.e. they do not separate. It is essential to emphasize that the intermolecular interactions between wax (typically long-chain hydrocarbons or esters and the like) and plasticizer molecules must be carefully balanced within an intermediate state, neither excessively strong nor excessively weak, to achieve the desired macroscopic properties in the final material.[6]
Overly strong interactions (e.g., extensive hydrogen bonding networks or strong dipole–dipole associations) would lead to a highly cohesive, tightly packed matrix in which segmental mobility of the wax chains is severely restricted. This would result in a rigid, brittle material with reduced flexibility, poor low-temperature performance, and diminished capacity for stress dissipation, properties against effective plasticization.
Conversely, interactions that are too weak (limited to sparse or feeble dispersion forces) would fail to ensure sufficient thermodynamic compatibility between the components. This could lead to phase separation, exudation (bleeding) of the plasticizer, macroscopic inhomogeneity, and loss of long-term performance stability. The optimal scenario involves a combination of intermolecular forces of differing strengths:
- Predominant weak, non-specific van der Waals (London dispersion) forces that are easily disrupted by thermal motion (ambient or processing temperatures).
- A controlled, moderate number of stronger but still reversible polar interactions, such as dipole–dipole attractions, induced dipole interactions, or a limited degree of hydrogen bonding (typically 0.5–2 hydrogen bonds per plasticizer molecule, depending on the size of the molecule and the system).[7]
These stronger interactions should be localized on specific functional moieties of the plasticizer (e.g., ester carbonyls, hydroxyls, ether oxygens, or aromatic rings) that can transiently associate with complementary electron-rich or electron-deficient sites on the wax molecules (oxygen atoms in ester waxes, terminal hydroxyls in fatty alcohols, etc.), hydrogen bonds being a good example.
The association must remain dynamic and reversible. The plasticizer molecules should undergo continuous attachment–detachment cycles. This constant exchange would allow individual plasticizer molecules to act as temporary “spacers” and lubricants: they momentarily shield chain–chain contacts, reduce frictional drag during segmental motion, and lower the energy barriers for conformational transitions, yet they are readily displaced by neighboring plasticizer molecules or by thermal fluctuations resulting in a microscopically heterogeneous but macroscopically homogeneous matrix with enhanced free volume and markedly improved flexibility and ductility.[8]
Consequently, the molecular design parameters of the plasticizer, molecular weight and architecture (linear vs. branched), polarity and distribution of functional groups, steric hindrance, conformational flexibility, and the precise balance between non-polar alkyl chains and polar head/tail groups, become paramount. Subtle variations in these parameters can shift the system from an optimally dynamic, compatible state to either an over-stabilized rigid network or an incompatible two-phase mixture. Rational selection, often aided by chemistry considerations and experimental validation (DSC, DMA, rheological studies), is therefore indispensable for achieving the delicate equilibrium required for high-performance wax-based formulations.[9]
Summarizing the benefits of plasticizers in a more practical manner in personal care, they accomplish:
Improved Flow Properties and Processability: The incorporation of these additives significantly enhances the rheological profile of the formulation. By reducing the yield stress and apparent viscosity at processing shear rates, the system becomes markedly easier to mix, pump, and fill during manufacturing. This improvement in flowability not only streamlines large-scale production but also facilitates precise dosing in consumer packaging. Additionally, the optimized rheology contributes to superior emolliency on skin, as the reduced internal friction allows the product to spread more readily and form a thinner, more uniform occlusive layer.
Superior Sensory Profile and Texture: The resulting microstructure* imparts a luxuriously smooth, creamy, and velvety tactile experience. This is achieved through the formation of a finely dispersed crystalline network or lamellar gel phase* that minimizes grittiness and maximizes perceived richness. Consumer perception studies consistently correlate such refined textures with premium quality and enhanced skin-conditioning benefits.
*Oleogels, frequently occur in personal care applications, liquids (oils) immobilized by a 3-D network of structuring agents, resulting in unique properties.
Enhanced Physical and Chemical Stability: The robust three-dimensional network created by the structuring system effectively immobilizes the liquid oil phase, dramatically reducing the incidence of syneresis, phase separation, and oil bleed over time and across temperature fluctuations. Simultaneously, it inhibits Ostwald ripening and polymorphic transitions in crystalline components, thereby preventing graininess, grittiness, or flaking.
Increased Mechanical Flexibility and Structural Integrity: In solid and semi-solid formats such as stick products, anhydrous balms, or transdermal patches, the structuring agents impart controlled plasticity and elasticity to the matrix. This modification significantly reduces brittleness and hardness, lowering the modulus of elasticity and increasing resistance to fracture under bending or compressive stress. Consequently, products exhibit minimal cracking, chipping, or flaking during application, removal from packaging, or normal consumer handling, thereby improving both durability and user acceptance.
Prolonged Fragrance Retention and Olfactory Performance: By entrapping volatile perfume molecules within the lipophilic crystalline or gel network, these structurants markedly slow the rate of evaporative loss. This is especially valuable in fine-fragrance formulations and deodorant
products.
Optimized Active and Functional Ingredient Distribution: The homogeneous microstructure ensures uniform dispersion and spatial stabilization of suspended particles, pigments, and functional actives throughout the bulk and at the product–skin interface. This is especially critical in photoprotective formulations (sunscreens), where even distribution of UV filters is essential for forming a continuous, uniform protective film, therefore, improved photometric uniformity, reduced hot spots of low protection, and consequently higher and more reproducible sun protection factors across the applied skin surface.
EXPERIMENTAL
Studies were conducted to complement the above detailed scientific arguments, and to summarize the benefits of Kester Wax K-70P, a new plasticizer from Koster Keunen, Inc. (INCI NAME: C18-38 Alkyl Hydroxystearoyl Stearate). In particular, the relevant crystallization and rheological properties that were investigated and correlated with the desired properties of an efficient plasticizer. By understanding the century-and-a-half evolution from camphor-plasticized Celluloid to today’s sophisticated cosmetic molecules, formulators can more rationally select plasticizers that deliver not only manufacturing ease but also measurable improvements in skin hydration, barrier function, and, most importantly, consumer-perceived luxury.
Materials, Methods and Instrumentation
Wax samples were prepared from Koster Keunen, Inc. commercial grade wax products with 5 % w/w of plasticizer unless listed otherwise. Crystallization studies were carried out using an Olympus CH microscope using polarized light. Images were captured using a Moticam 3 3.0 MP camera integrated with the microscope. Microscope slides and cover glasses were purchased from Fisher Scientific and were electrically heated before cooling to ambient temperature for inspecting the crystallization process and the resulting crystalline particles.
Dynamic stress sweep rheological testing and analysis were performed at the Institute of Material Science at the University of Connecticut, Storrs. Special acknowledgement to Dr. Dennis Ndaya for his collaboration and expertise. A Discovery HR20 rheometer (DHR20, TA) was employed utilizing a serrated Peltier plate fixture. The test configuration featured a modified cross-hatched 40 mm cone plate. Samples were loaded and zero gap determination was performed at the designated test temperature. A Peltier temperature control system connected to a recirculating water bath maintained the system at the required temperature throughout the tests. Yield stress measurements were performed using dynamic stress sweep experiments at 25 °C and 6.28 rad/s. The yield stress was determined from the cross over point (G′ = G′′).
Crystallization Behavior of Natural Products
As previously stated, the crystallization process of natural waxes is influenced by factors such as temperature, cooling rate, and the presence of extraneous materials, e.g. impurities or additives. Typically, slow cooling results in the formation of larger, more well-defined crystals, whereas rapid cooling tends to produce smaller, less organized structures. Close attention to the crystallization behavior can enhance the performance and stability of products incorporating natural plant waxes.
Plasticizing is accomplished by the plasticizer molecules embedded in the wax matrix between the molecules. Besides the Thermodynamic or Mechanistic Theories (discussed above by the molecular movement of attachment–detachment cycles), the Free-Volume Theory explains that additional volume is created between the wax molecules.[10] This is evident by examining the branched chain configuration in Kester Wax K-70P (Fig. 1.).
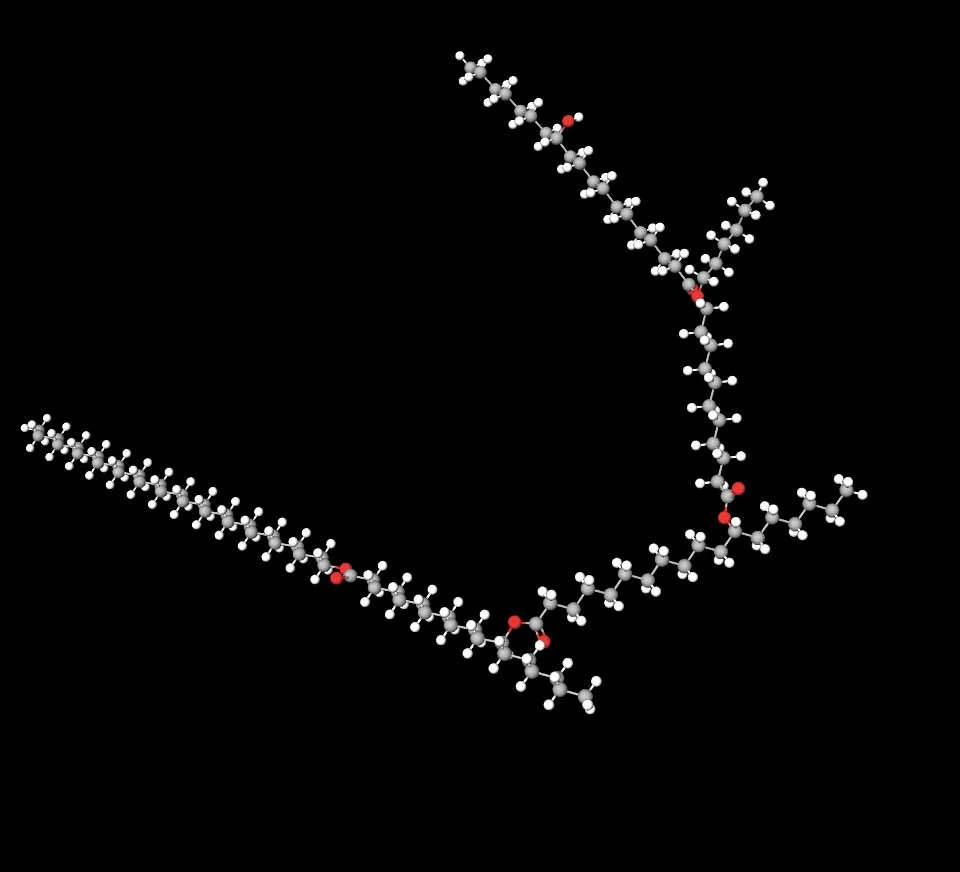
Figure 1. 3-D, Ball-and-Stick (Model) image showing the branched spacious structure of Kester Wax K-70P. Carbon atoms are depicted in grey, hydrogen atoms in white and oxygen atoms in red.
The embedded plasticizer molecules will also provide more separation between the wax molecules weakening the interaction between them (Gel Theory) and providing some lubricity (Lubricity Theory).[11] These will modify the crystallization characteristics, alter the crystallization kinetics and crystal morphology. The result is a more flexible and pliant wax matrix with a uniform distribution of smaller crystals throughout the material. This reduces the materials’ brittleness, i.e. enhances their ability to withstand higher stress and deformation without fracturing, while maintaining the necessary protective and moisturizing properties. Understanding and controlling this crystallization behavior is essential for creating high-quality, effective personal care products that meet consumer expectations.
Building upon our prior discussion regarding the pivotal role of plasticizers in conferring emolliency through enhanced molecular mobility and flexibility within topical formulations, it is instructive to extend this analysis to the two complementary mechanistic pillars of skin moisturization: occlusivity and humectancy.
The molecular-level uniformity previously elucidated, arising from the plasticizer’s capacity to disrupt inter-chain crystalline domains and promote a homogeneous amorphous phase, directly translates into the formation of a continuous, defect-free lipid film upon application. This macroscopic homogeneity manifests as a highly coherent and impermeable barrier on the stratum corneum surface, significantly reducing transepidermal water loss (TEWL).[3] Such behavior aligns precisely with the classical definition of an occlusive moisturizing agent, wherein physical impediment of water evaporation is achieved through the establishment of a durable hydrophobic layer, thereby preserving cutaneous hydration over extended periods.
Concurrently, examination of the Kester Wax K-70P molecule (as depicted in Figure 1) reveals structural features that confer intrinsic humectant properties independent of its primary plasticizing function. The presence of multiple electronegative oxygen atoms within the ester and ether linkages endows Kester Wax K-70P with strong hydrogen-bond acceptor capability. These electron-rich sites readily form energetically favorable hydrogen bonds with water molecule, enabling the molecule to attract, immobilize, and retain water within the upper layers of the stratum corneum even under low relative humidity conditions.[4] This water-binding capacity operates via a classic humectant mechanism, distinct from mere occlusion, and contributes additively to overall skin hydration by establishing a hydrated microenvironment that counteracts desiccation stresses.
Thus, Kester Wax K-70P exemplifies a rare multifunctional excipient that simultaneously enhances film uniformity (> superior occlusivity), reduces brittleness through plasticization (> sustained emolliency), and actively sequesters water through hydrogen-bonding interactions (> measurable humectancy), resulting in a tripartite moisturization profile that can be greater than the sum of its individual contributions
RESULTS AND DISCUSSION
Crystallization Behavior Through PLM
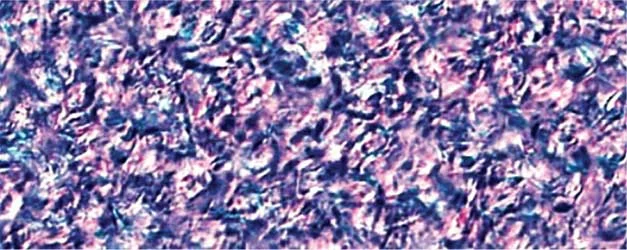
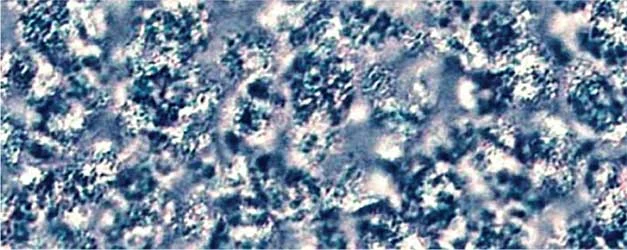
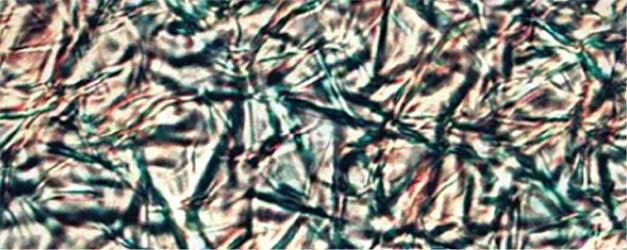
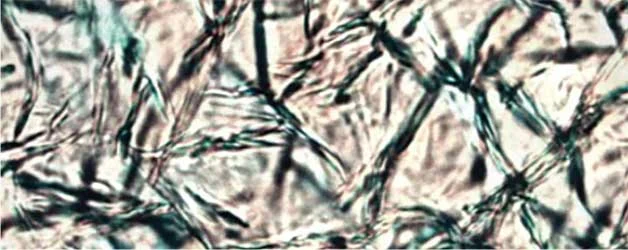
Figure 2. PLM Crystallization Photos at STP of various Natural Waxes at 10% w/w in Caprylic Capric Triglyceride without and with Kester Wax K-70P
Close examination of Kester Wax K-72 (Behenyl Behenate) reveals a more segmented crystal structure with smaller crystal sizes when K-70 plasticizer is added, consistent with the suppression of crystal formation.
With natural Waxes, such as Sunflower Wax and Carnauba Wax as well as with the naturally derived, medium chain ester wax stearyl behenate (Kester Wax K-62) the plasticizing effect is manifested differently, i.e. large spaces between the crystals providing elasticity to the matrix and decreasing the cohesive forces between the crystals formed. This becomes more evident by rationalizing the rheometry studies discussed below.
Flow Properties with Kester Wax K-70P Plasticizer
Rheology, being the study of flow and deformation properties of a wide range materials, is a convenient and valuable tool for determining the effect of plasticizers on these physical properties of wax matrices. We have compared the All Over Body Balm formulas (composition details can be found in the formula section below) containing both no plasticizer and 15 % Kester Wax K-70P in Dynamic Stress Sweep tests. At low stress amplitudes, both the elastic G′ and the viscous G′′ modulus formed a constant plateau region known as the linear viscoelastic range (LVR) of the materials. In the LVR, the stress induced in response to imposed strain occurs in proportionality and, therefore, a linear range is found. For all studied body balms, G′ was observed to be higher than G′′ in the LVR, revealing a dominating elastic character of the products at low stress range. The greater the elastic nature of the products leads to a substantial elongation of the structural network under continually increasing straining/stress force before the structure is distorted into a state of viscoelastic liquid.
Base Formula (No Plasticizer) Base Formula +15% Kester Wax K-70P Modulus (Pa) Stress (Pa) Modulus (Pa) Stress (Pa) 155.5 45581.9 131.9 44644.5
| Plasticizer Function | Mechanistic Effect | ||
| Modulus (Pa) | Stress (Pa) | Modulus (Pa) | Stress (Pa) |
| 155.5 | 45581.9 | 131.9 | 44644.5 |
Table 1. Modulus and Stress values for All Over Body Balm in absence and presence of plasticizer.
When plasticizer is present, it becomes evident how plasticizing waxes help to inhibit crystallization, which may not be essential for the product’s overall structure but instead plays a crucial role in enhancing thermal stability and overall formula integrity. The same All Over Body Balm formula with 15% added oils and no plasticizer also shows that the formula’s integrity is compromised at an earlier stage of stress, indicating that it may not maintain its structure at elevated stability temperatures due to the lack of appropriate crystal formation.
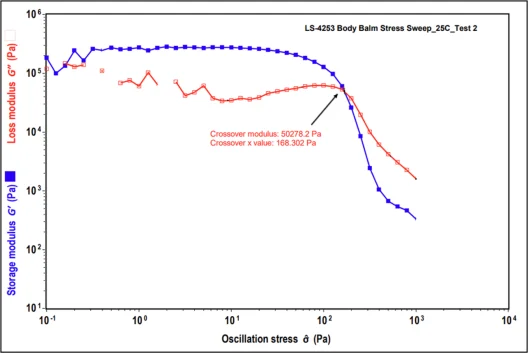
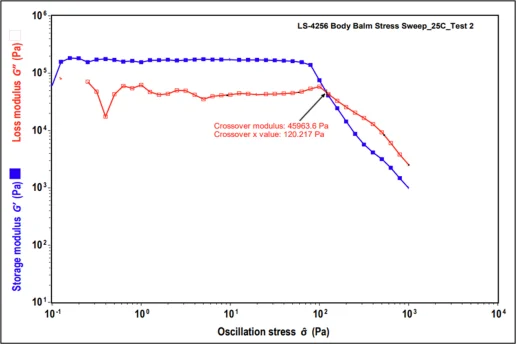
Figure 2. Dynamic Stress Sweep results for All Over Body Balm in absence and presence of plasticizer.
Formulation Results
Formula 1: Squeezable Lip Balm – with 5% K-70P – Most popular formats containing C18-38 Alkyl Hydroxystearoyl Stearate
| Trade Name | INCI Name | % |
| Phase A | ||
| Sunflower Wax | Helianthus Annuus (Sunflower) Seed Wax | 5.0 |
| Kester Wax K-70P | C18-38 Alkyl Hydroxystearoyl Stearate | 5.0 |
| Coconut Oil | Cocos Nucifera (Coconut) Oil | 30. |
| Jojoba Oil | Simmondsia Chinensis (Jojoba) Seed Oil | 30.0 |
| Jeechem CTG | Caprylic/Capric Triglyceride | 30.0 |
Formula 2: All Over Body Balm – Comparison Formulas – Most popular format containing C18-38 Alkyl Hydroxystearoyl Stearate
| Trade Name | INCI Name | BASE | OILS | With Plasticizer |
| Phase A | ||||
| AR Cocoa Butter | Theobroma Cacao (Cocoa) Butter | 14.1 | 12.0 | 12.0 |
| Sunflower Wax | Helianthus Annuus (Sunflower) Seed Wax | 8.8 | 7.5 | 7.5 |
| *Plasticizer | — | — | 15.0 | |
| Kester Wax K-24 | Lauryl Laurate | 9.4 | 8.0 | 8.0 |
| Extra Virigin Coconut Oil | Cocos Nucifera (Coconut) Oil | 40.0 | 49.0 | 34.0 |
| Jojoba Oil | Prunus Amygdalus Dulcis (Sweet Almond) Oil | 15.9 | 13.5 | 13.5 |
| Rice Bran Oil | Oryza Sativa (Rice Bran) Oil | 10.6 | 9.0 | 9.0 |
| Vitamin E Acetate | Tocopheryl Acetate | 1.2 | 1.0 | 1.0 |
CONCLUSION
In line with the objectives of this article we have investigated the efficiency of Kester Wax K-70P for changing the morphological and rheological properties of certain wax matrices. These properties, modification of these properties, are crucial for a plasticizer. The experimental results support the requirements described in the scientific literature. By modifying the crystallization and flow/ deformation properties of the wax matrices subjected to this research, Kester Wax K-70P, our assessment is that this material is a promising addition to the plasticizer market. Since it is globally accepted and made from naturally sourced ingredients, it will greatly benefit the users, formulator in the personal care industry where improved workability and quality is desired.
REFERENCES
[1] Hyatt, J. W. (1869). U.S. Patent No. 105,338. Improvement in the manufacture of celluloid.
[2] Idson, B. (1992). Dry skin: moisturizing and emolliency. Cosmetics & Toiletries, 107(7), 69–78.
[3] Rawlings, A. V., & Harding, C. R. (2004). Moisturization and skin barrier function. Dermatologic Therapy, 17(s1), 43–48. https://doi.org/10.1111/j.1396-0296.2004.04s1005.x
[4] Lodén, M. (2003). The role of topical humectants in the skin. American Journal of Clinical Dermatology, 4(11), 771–788. https://doi.org/10.2165/00128071-200304110-00006
[5] Wypych, G. (2017). Handbook of Plasticizers (3rd ed.). ChemTec Publishing.
[6] Sears, J. K., & Darby, J. R. (1982). The Technology of Plasticizers. Wiley-Interscience.
[7] Krause, S., & Roman, N. (1965). Hydrogen bonding in plasticized polymer systems. Journal of Polymer Science Part A: General Papers, 3(5), 1631–1643.
[8] Ferry, J. D. (1980). Viscoelastic Properties of Polymers (3rd ed.). John Wiley & Sons.
[9] Mezger, T. G. (2020). The Rheology Handbook (5th ed.). Vincentz Network.
[10] Fox, T. G., & Flory, P. J. (1950). Second-order transition temperatures and related properties of polystyrene. Journal of Applied Physics, 21(6), 581–591.
[11] Boyer, R. F. (1963). The relation of transition temperatures to chemical structure in high polymers. Rubber Chemistry and Technology, 36(5), 1303–1421.


